|
If a list were made of all elements, we would find the sequence halogen, noble gas, alkali metal, and alkaline-earth metal several more times.ĭmitri Ivanovich Mendeleev proposed the periodic law behind his periodic table compiling. Exactly the same sequence is repeated eight elements later (Ne, Na, and Mg), but this time a halogen (F) precedes the noble gas. The second, third, and forth elements on the list (He, Li, and Be) are a noble gas, an alkali metal, and an alkaline-earth metal, respectively. 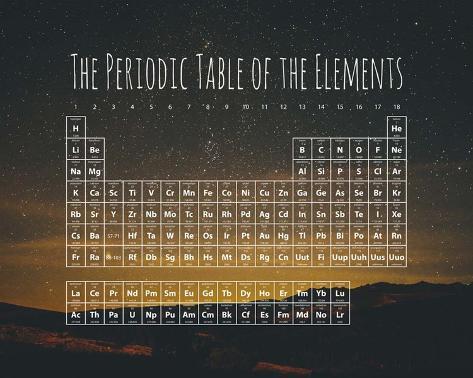
Obtaining atomic weights, we haveĮlements which belong to families we have already discussed are indicated by shading around their symbols. You can see that such a relationship exist by listing symbols for the first dozen elements in order of increasing relative mass. Therefore it seems reasonable to expect some correlation between this microscopic property and macroscopic chemical behavior. This could account for the related chemical reactivities and analogous compounds of these elements.Īccording to Dalton’s atomic theory, different kinds of atoms may be distinguished by their relative masses (atomic weights). Atoms of sodium ought to be similar in some way to atoms of lithium, potassium, and the other alkali metals. The similarities among macroscopic properties within each of the chemical families lead one to expect microscopic similarities as well.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
